Patient Presentation
A 79-year-old male patient in good general health presented with gradually declining vision and increasing difficulty reading. He was physically active with no significant medical history. His ocular history included bilateral pseudophakia OU and a prior pars plana vitrectomy with membrane peel OD for an epiretinal membrane in 2020.
Diagnosis
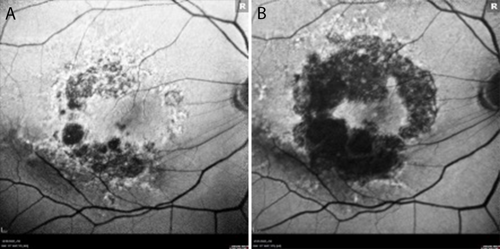
At baseline, best-corrected visual acuity (BCVA) was 20/40 in each eye. Color fundus photography (Figure 1) and fundus autofluorescence (FAF) imaging (Figure 2) revealed bilateral geographic atrophy (GA) secondary to dry age-related macular degeneration (AMD). The right eye demonstrated a moderate multifocal nonsubfoveal GA with hyperautofluorescent borders, a hallmark of active progression. The left eye showed large and numerous central drusen with pigmentary changes and only very mild atrophy. No choroidal neovascularization was present in either eye.
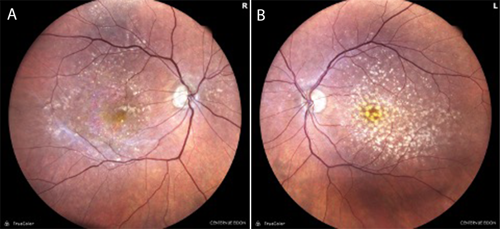
Treatment
Given the markedly asymmetric disease, I initiated treatment with an FDA-approved intravitreal complement inhibitor in the right eye in November 2023, where nonsubfoveal GA with active progression posed the most immediate threat to central vision. Injections were continued every other month. At that time, the left eye had only minimal atrophy and did not warrant intervention. Approximately 2 years later, in April 2025, imaging revealed early central GA OS. Given the patient’s strong desire to preserve vision and the likelihood of foveal involvement within his lifetime, I initiated treatment in the left eye as well.
Outcomes
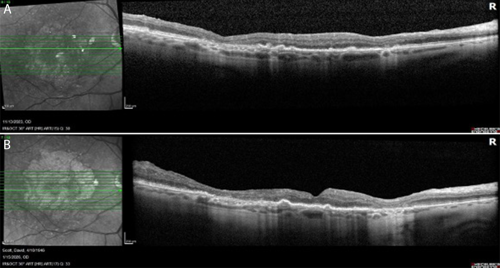
Throughout more than 2 years of treatment OD and 9 months OS, VA has remained stable at 20/40 bilaterally. FAF imaging at approximately 2 years demonstrates continued GA enlargement OD, though with preservation of a central foveal island (Figure 2b). OCT confirms stable foveal architecture despite peripheral atrophic progression (Figure 3). No adverse events have been observed, and the patient continues his active lifestyle.
Discussion
This patient exemplifies the profile that benefits most from complement inhibition for GA. Several factors made him an ideal candidate. First, his right eye had nonsubfoveal GA with a preserved foveal island, and he started treatment while the lesion was progressing but had not yet reached the fovea captured the optimal therapeutic window. Second, deferring treatment OS until early central GA emerged demonstrated how monitoring can identify the right moment for intervention in asymmetric disease. When foveal-threatening changes appeared, treatment was initiated promptly. Third, his commitment to long-term injections was essential, as clinical trial data show that complement inhibition benefit increases with sustained treatment. Patients treated for 36 months showed a cumulative reduction in lesion growth of approximately 20% to 30%.1,2
Notably, the patient’s prior vitrectomy did not appear to diminish a treatment response over 2 years. This case also illustrates that bilateral GA does not always require simultaneous treatment. When disease is markedly asymmetric, staging by severity and initiating in the fellow eye when progression warrants it is a rational approach.
The ideal candidate for complement inhibitor therapy presents with nonsubfoveal GA, preserved central vision at risk of decline, and commitment to long-term treatment. Identifying and treating such patients early—before the window of foveal preservation closes—remains the most impactful decision a retina specialist can make in managing geographic atrophy.
References
- Heier JS, Lad EM, Holz FG, et al. Pegcetacoplan for the treatment of geographic atrophy secondary to age-related macular degeneration (OAKS and DERBY): two multicentre, randomised, double-masked, sham-controlled, phase 3 trials. Lancet. 2023;402(10411):1434-1448. doi:10.1016/S0140-6736(23)01520-9
- Wykoff CC, Holz FG, Chiang A, et al; OAKS, DERBY, and GALE Investigators. Pegcetacoplan treatment for geographic atrophy in age-related macular degeneration over 36 months: data from OAKS, DERBY, and GALE. Am J Ophthalmol. 2025;276:350-364. doi:10.1016/j.ajo.2025.04.016
This content is sponsored by Apellis









